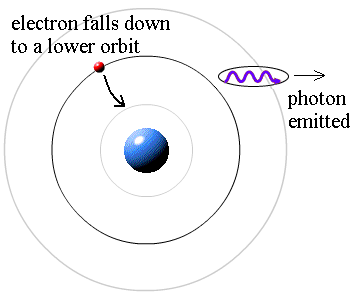
PDF) Shape and size of electron are determine by the amount of photon energy incidence on it 5e7f7e40d7564 (1) | Saddam H U S A I N Dhobi - Academia.edu
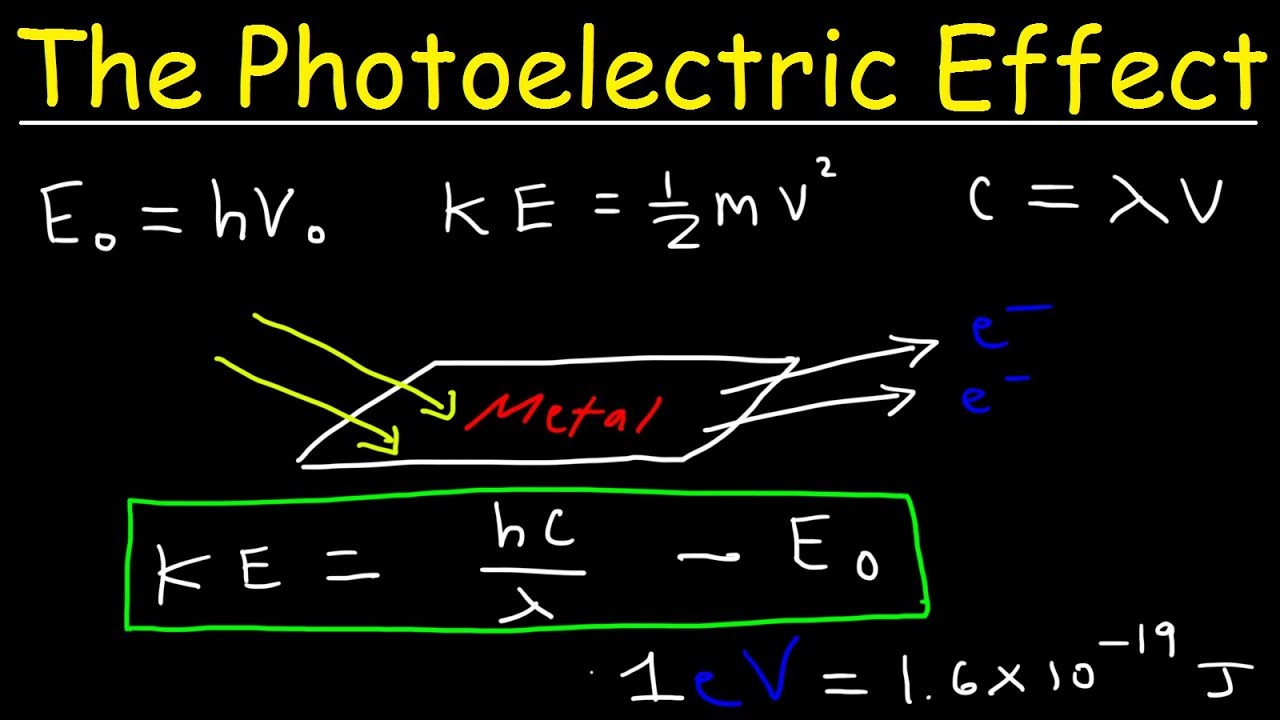
Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr - YouTube

CHEM 101: Photoelectric Effect - Threshold Frequency and Binding Energy from Kinetic Energy - YouTube

Coupling between Harmonic Vibrations Influences Quantum Beating Signatures in Two-Dimensional Electronic Spectra | The Journal of Physical Chemistry C
I have a question where I have to find out the maximum kinetic energy from the wavelength, frequency of photon and stopping potential. How do I find the maximum kinetic energy from

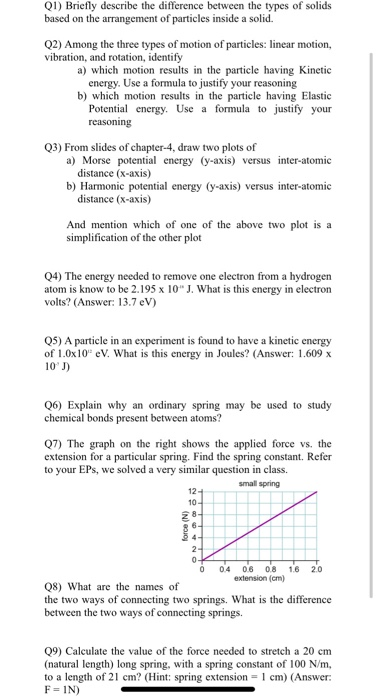
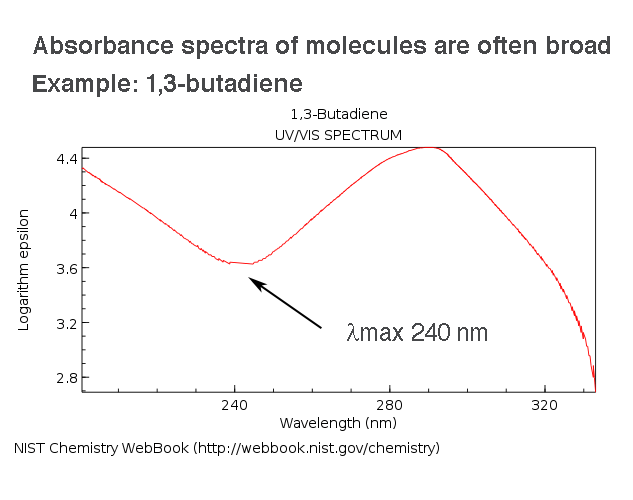
About these slides These slides are used as part of my lessons and shouldn't be considered comprehensive There's no excuse for not turning up to lessons! - ppt video online download