SOLVED: 11.50 Hydrogen peroxide, H2O2, has a normal boiling point of 150°C. Based on the data given in Figure 11.25, would you expect hydrogen peroxide to have a higher or lower pressure
Sciencemadness Discussion Board - Hydrogen Peroxide - Illustrated Practical Guide - Powered by XMB 1.9.11

fuel - If high-test peroxide is most stable when pure, why are most uses of it in rocketry at lower concentrations? - Space Exploration Stack Exchange

Ph Level 3.5 Liquid Hydrogen Peroxide Used For Industrial, Density 1.45 G/Cm Boiling Point: 150.2 I? C at Best Price in Delhi | Kushal Chemicals

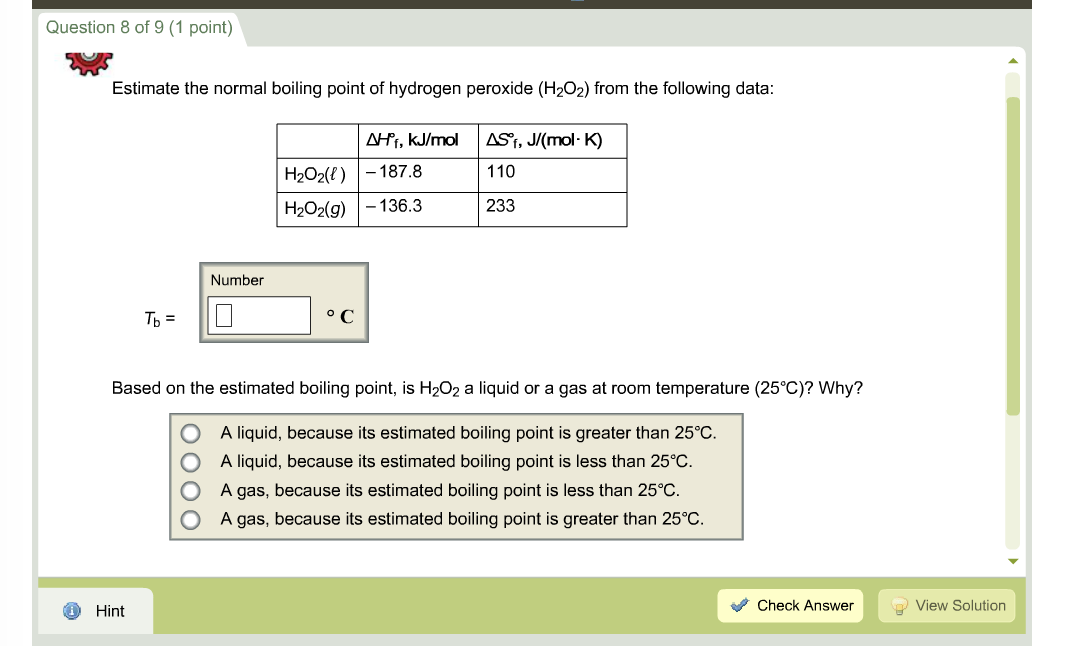
SOLVED:Direct measurement of the normal boiling point of hydrogen peroxide is not possible because pure H2 O2 explodes on heating. The boiling point can be estimated, however, from vapor-pressure data. Use the